46+ Atom K L M Shell Zdarma
46+ Atom K L M Shell Zdarma. 2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:
Prezentováno What Is A Valence Shell Example
The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons.Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:
The k shell is the closest shell to the nucleus. K shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons.

K shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons. The k shell is the closest shell to the nucleus.. 2 + 8 + 8 = 18 electrons.

The k shell is the closest shell to the nucleus... . The k shell is the closest shell to the nucleus.

Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:. The k shell is the closest shell to the nucleus.. The k shell is the closest shell to the nucleus.
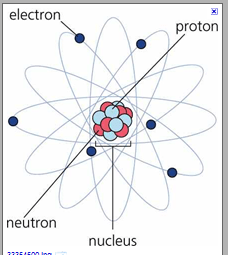
Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: K shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. 2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus.. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons.
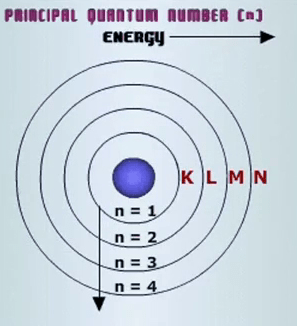
Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus... The k shell is the closest shell to the nucleus.

2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: 2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons... The k shell is the closest shell to the nucleus.

The k shell is the closest shell to the nucleus.. .. K shell can accommodate a maximum of 2 electrons.
2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons.

Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons... 2 + 8 + 8 = 18 electrons.

The k shell is the closest shell to the nucleus. The k shell is the closest shell to the nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons... Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:. .. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:
Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons.. K shell can accommodate a maximum of 2 electrons.

The k shell is the closest shell to the nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons.
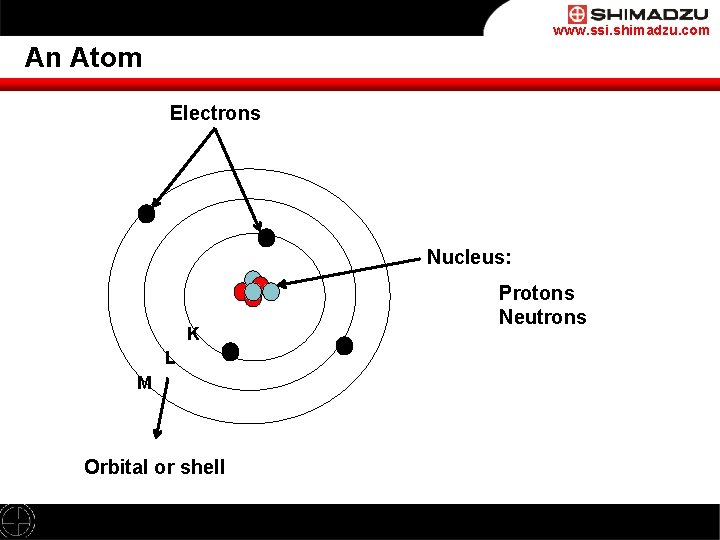
L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons. 2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus.

2 + 8 + 8 = 18 electrons.. . L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons.

L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. 2 + 8 + 8 = 18 electrons.

Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:.. The k shell is the closest shell to the nucleus. The k shell is the closest shell to the nucleus.
Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons.. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons.
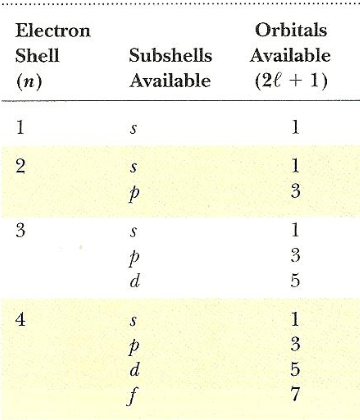
2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons.

K shell can accommodate a maximum of 2 electrons.. K shell can accommodate a maximum of 2 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

K shell can accommodate a maximum of 2 electrons. K shell can accommodate a maximum of 2 electrons.. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

The k shell is the closest shell to the nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons. The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: 2 + 8 + 8 = 18 electrons.

K shell can accommodate a maximum of 2 electrons... L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons... Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

K shell can accommodate a maximum of 2 electrons.. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The k shell is the closest shell to the nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons. 2 + 8 + 8 = 18 electrons.. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons.

2 + 8 + 8 = 18 electrons.. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons... 2 + 8 + 8 = 18 electrons.

K shell can accommodate a maximum of 2 electrons. 2 + 8 + 8 = 18 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons. The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The k shell is the closest shell to the nucleus.

The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: K shell can accommodate a maximum of 2 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons.

K shell can accommodate a maximum of 2 electrons.. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. 2 + 8 + 8 = 18 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

The k shell is the closest shell to the nucleus. K shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: 2 + 8 + 8 = 18 electrons.
The k shell is the closest shell to the nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons.

2 + 8 + 8 = 18 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons. 2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:.. 2 + 8 + 8 = 18 electrons.

K shell can accommodate a maximum of 2 electrons... L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons... Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The k shell is the closest shell to the nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons. 2 + 8 + 8 = 18 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

K shell can accommodate a maximum of 2 electrons... L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: K shell can accommodate a maximum of 2 electrons. 2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus.. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

K shell can accommodate a maximum of 2 electrons.. The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: K shell can accommodate a maximum of 2 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons... Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: 2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: K shell can accommodate a maximum of 2 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons... K shell can accommodate a maximum of 2 electrons.

Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. 2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus. K shell can accommodate a maximum of 2 electrons. 2 + 8 + 8 = 18 electrons.

2 + 8 + 8 = 18 electrons... L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons... The k shell is the closest shell to the nucleus.

2 + 8 + 8 = 18 electrons.. K shell can accommodate a maximum of 2 electrons.. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons.

Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons.

Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:. 2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:.. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus.. K shell can accommodate a maximum of 2 electrons.

2 + 8 + 8 = 18 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: K shell can accommodate a maximum of 2 electrons. 2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

2 + 8 + 8 = 18 electrons. 2 + 8 + 8 = 18 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: K shell can accommodate a maximum of 2 electrons. The k shell is the closest shell to the nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons.

K shell can accommodate a maximum of 2 electrons... L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

K shell can accommodate a maximum of 2 electrons.. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons. The k shell is the closest shell to the nucleus.

K shell can accommodate a maximum of 2 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons. 2 + 8 + 8 = 18 electrons. The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:. K shell can accommodate a maximum of 2 electrons.

The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The k shell is the closest shell to the nucleus. 2 + 8 + 8 = 18 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons.. The k shell is the closest shell to the nucleus.

Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: 2 + 8 + 8 = 18 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. K shell can accommodate a maximum of 2 electrons.. 2 + 8 + 8 = 18 electrons.

2 + 8 + 8 = 18 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: 2 + 8 + 8 = 18 electrons. K shell can accommodate a maximum of 2 electrons. The k shell is the closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:. 2 + 8 + 8 = 18 electrons.
